Multiple sclerosis (MS) is a chronic inflammatory demyelinating and neurodegenerative disease affecting the central nervous system. Interferon (IFN) β-1a 44 µg, dimethyl fumarate (DMF) and fingolimod are established drugs for the treatment of relapsing-remitting MS (RR MS). The aim of this project, which includes analysis of data from the ReMuS registry, was to compare the efficacy of IFN β-1a 44 µg, DMF and fingolimod in patients with RR MS in real-world clinical practice settings in the Czech Republic.

Pavelek, Z., Sobíšek, L., & Vališ, M. (2017). A comparison of efficacy of subcutaneous interferon β-1a 44 μg, dimethyl fumarate and fingolimod in the real-life clinical practice - a multicenter observational study. In Czech and Slovak Neurology and Neurosurgery (Vol. 81/114, Issue 4, pp. 457-465). Care Comm. https://doi.org/10.14735/amcsnn2018457
Abstract
Introduction: Multiple sclerosis is a chronic inflammatory and neurodegenerative disease affecting the CNS. Established drugs for relapsing-remitting MS (RR MS) include interferon (IFN) β-1a 44 μg, dimethyl fumarate (DMF) and fingolimod. The aim of the project, or the outcome of the analysis from the ReMuS registry, was to compare the efficacy of subcutaneously administered IFN β-1a 44 μg, DMF and fingolimod in patients with RR RS who were started on this treatment within 90 days of relapse onset in real clinical practice in the Czech Republic.
File and methodology: A total of 279 patients with RR MS who experienced one relapse during first-line treatment (IFN β-1a 22 μg 3× weekly, IFN β-1a 30 μg 1× weekly, IFN β-1b 250 μg obden, teriflunomide 14 mg daily, glatiramer acetate 20 mg daily or glatiramer acetate 40 mg 3× weekly) and who were switched to either IFN β-1a 44 μg or DMF or fingolimod were included. Parameters monitored were the annualized relapse rate (ARR), time to next relapse, proportion of patients without relapse, and change in Expanded Disability Status Scale (EDSS) one year after treatment change for each agent.
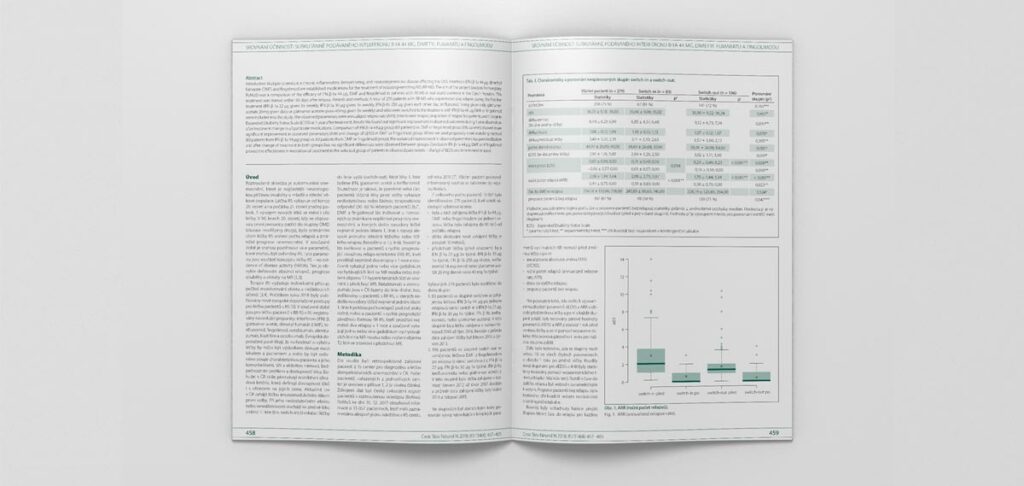
Results: After the change of therapy, all three investigated preparations showed a significant improvement of the analysed parameters during the one-year observation period. When comparing the group of patients treated with IFN β-1a 44 μg (83 patients) vs. fingolimod or DMF (196 patients), there was a more significant improvement in the ARR parameter and change in EDSS in the latter group. After pairing patients on IFN β-1a 44 μg therapy with the DMF or fingolimod-treated group in a 1 : 1 ratio by propensity score matching (83 vs. 83 patients), there remained a significant effect on the improvement of the studied parameters before and after the change in therapy in both groups, but we no longer demonstrated a significant difference in effect between the groups.
Conclusion: IFN β-1a 44 μg, DMF and fingolimod proved their efficacy in escalation therapy in a selected group of patients in the parameters of change in EDSS and time to next relapse.
The full article is available in Czech by logging in to the journal's website Czech and Slovak Neurology and Neurosurgery.