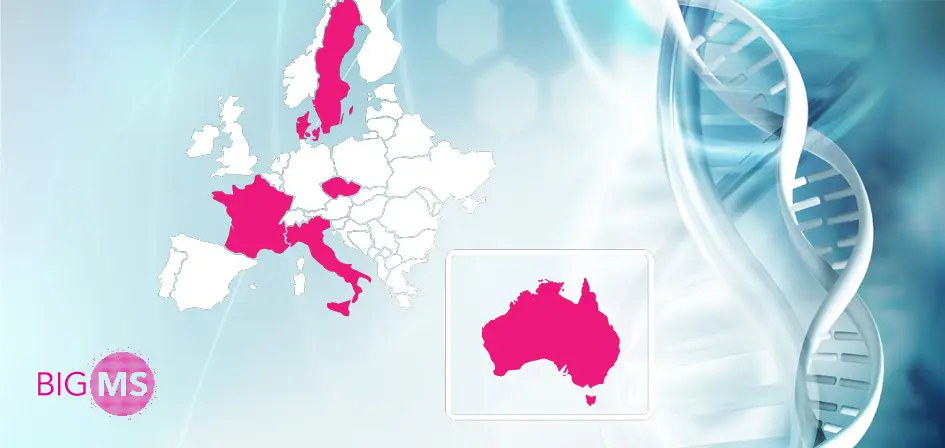
Article from Assoc. Prof. Anna Glaser from Karolinska Institutet, Sweden, focuses on the importance and contribution of European multiple sclerosis registries, specifically the Big Multiple Sclerosis Data Network (BMSD) project, in which ReMuS is actively involved. In it, readers will find an overview of what data is being collected across Europe and how this data is helping not only scientists but also physicians to improve patient care.

Glaser, A., Butzkueven, H., van der Walt, A., Gray, O., Spelman, T., Zhu, C., Trojano, M., Iaffaldano, P., Battaglia, M. A., Lucisano, G., Vukusic, S., Vukusic, I., Casey, R., Horakova, D., Drahota, J., Magyari, M., Joensen, H., Pontieri, L., Elberling, F., Klyve, P., Mouresan, E.F., Forsberg, L., Hillert, J. (2024). Big Multiple Sclerosis Data network: an international registry research network. And Journal of Neurology (Vol. 271, Issue 6, pp. 3616-3624). Springer Science and Business Media LLC. https://doi.org/10.1007/s00415-024-12303-6 PMID: 38561543
Abstract
Introduction: Big Multiple Sclerosis Data (BMSD) Network (https://bigmsdata.org) was established in 2014 and includes national multiple sclerosis (MS) registries from the Czech Republic, Denmark, France, Italy and Sweden, as well as the international MSBase registry. The BMSD has addressed the ethical, legal, technical and administrative challenges of data sharing and has so far published three scientific articles based on collaborative datasets as a proof of concept for its collaborative model.
Data collection: Although BMSD registries operate independently on different platforms, similarities in variables, definitions and data structure allow for common analyses. Following the coordinated decisions of the BMSDs, some changes in the collection of adverse event data have been implemented, demonstrating the ability of these registries to co-evolve.
Data management: Scientific projects can be proposed by external sponsors through the Coordination Centre and each Registry decides independently whether to participate, respecting its administrative structure. The research datasets are created on a project-by-project basis, with a specific data model developed for each project, based on a single underlying model. To overcome data sharing challenges, BMSD has developed procedures for federated data analysis.
Future perspectives: BMSD is currently seeking a qualifying opinion from the European Medicines Agency (EMA) to conduct post-authorisation safety studies (PASS) and plans to seek qualification for post-authorisation efficacy studies (PAES) as well. BMSD aims to support the development of research based on real-world data in the field of multiple sclerosis.
You can read the full article in English on the magazine's website Journal of Neurology.